A pressure column, also called a manometer, is often used inside a laboratory, for meteorology applications, or in aviation so there is a limit on the height of any instrument. A dense liquid is helpful for pressure column applications because it minimizes the height needed for a column. Historically, pressures were measured in millimeters of mercury (mmHg) because mercury is the densest liquid at room temperature.

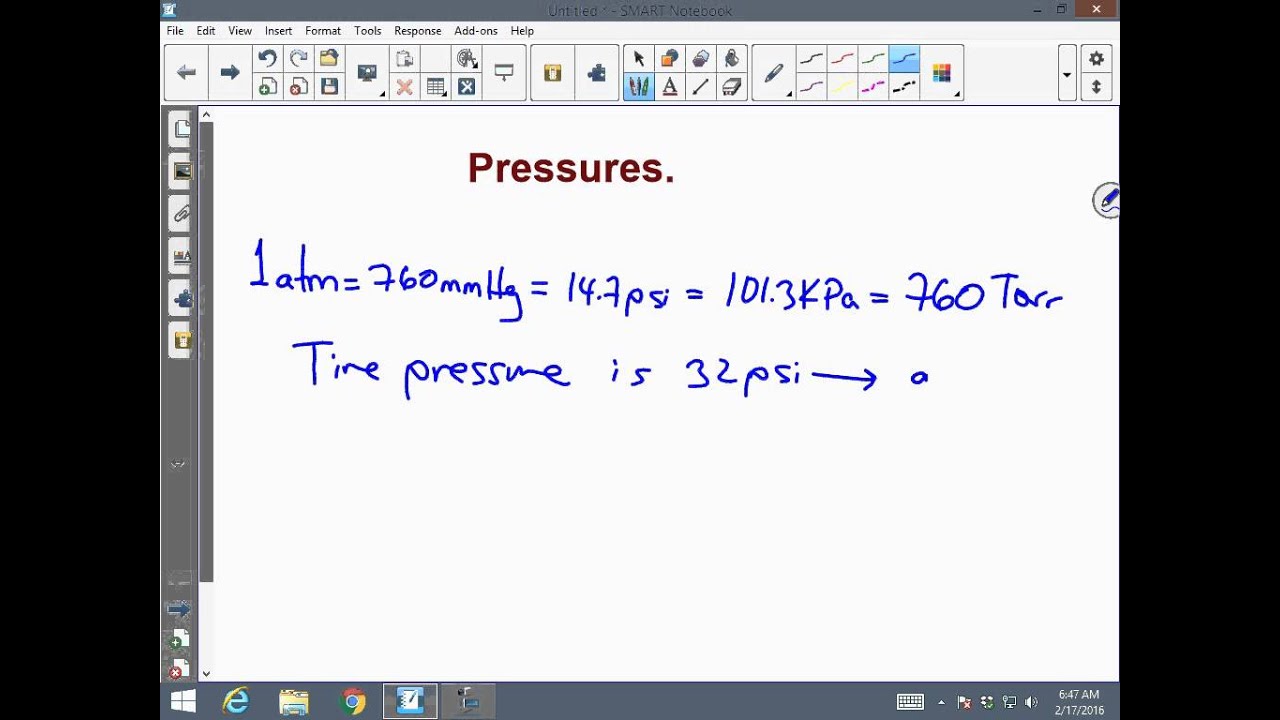
There are 760 mmHg in a standard atmosphere but only 0.0013157894736842 standard atmospheres (atm) in one mmHg. Yes, one standard atmosphere (atm) is greater than one millimeter of mercury (mmHg). Both approaches will give you the correct solution. The first option is to divide mmHg by 760, which is the number of mmHg in 1 atm. You have two conversion options to convert millimeters of mercury (mmHg) to standard atmospheres (atm). It is equivalent to the average atmospheric pressure on Earth at sea level. What is atm?Ī Standard atmosphere (atm) is a unit of pressure defined as 101,325 Pa, 760 mmHg, 1.01325 bar, 760 torr, 4.6959 psi, or 1 ata (atmosphere absolute). You will most frequently encounter mmHg from blood pressure readings and a meteorologist’s weather reports in everyday life. It is a manometric unit of pressure and is an abbreviation for millimeters of mercury.
MMHG TO ATM CONVERTER FOR ANDROID
Pressure = 48,000 Pa × 0.0075 = 360.02956ĭownload Toppr app for Android and iOS or signup for free.An mmHg is a unit of pressure that is defined as equal to the pressure exerted by a column of mercury that is one millimeter high under standard gravity. What is the pressure of mm Hg if Pa is 48000? Pressure = 36,000 Pa × 0.0075 = 270.02 mmHg Solved Question for You Now calculate the pressure of 36,000 Pa using this same formula from step three: The solution of this problem produces the formula:

Suppose 1 mmHg resembles to 1 Pa × 1,01,325 / 760 pressure (mm Hg) resembles to pressure (Pa). Now to covert pressure Pa using proportion the Formula is: Now multiply both side of the equation by 1 / 760 you would get: So, we can conclude that 1,01,325 Pa = 760 mmHg. Let’s consider 1 atm = 1,01,325 Pa, and 1 atm = 760 mmHg (As discussed earlier). Now we obtain the relation between 1 Pa and 1 mmHg. Pressure = Hg Density × Standard Gravity × Mercury Height Using the basic definition of mmHg calculate the blood pressure of 120 mm Hg. In addition, the mmHg is still widely used in the B.P. Moreover, the unit of a millimetre of mercury is considered obsolete and the S.I unit Pascal (Pa 1 atm = 1,01,325 Pa) ought to be used. And we also know that one mmHg is also equal to 1 torr, which is 1 / 760 of atmospheric pressure (atm) that is 1 atm = 760 mmHg.
MMHG TO ATM CONVERTER HOW TO
How to calculate mmHg?Īs discussed earlier we know that one millimetre of mercury is the pressure exerted by a 1mm vertical column of mercury at 0 degree Celsius. In addition, they displayed the pressure alteration among two fluids as a vertical variance between the mercury levels of two connected reservoirs. But, they are no longer in usage due to the toxicity of mercury.įurthermore, the sensitivity of mercury columns to temperature and local gravity and the better convenience of other instrumentation also reduce their use. Also, mercury manometer is the first and most accurate pressure gauge.
Moreover, these two units are not equal and the relative difference (less than 0.000015 per cent) is minor for most practical use. In addition, one millimetre of mercury is approximately 1 Torr which is 1 / 760 of standard atmospheric pressure (101325 / 760 ≈ 133.322368421053 Pascals).
In this the mm represents millimetre and Hg is the chemical name of mercury or the symbol for mercury.įurthermore, the millimetre of mercury is still routinely used in medicine, aviation, meteorology, and many other scientific fields. Moreover, we denote it with mmHg or mm Hg. Also, it is currently defined as exactly 133.3223 Pascals. Besides, one can describe it as the extra pressure whose production takes place by a column of mercury one millimetre high. It is a unit of measure that uses the manometric unit to tell the pressure of the objects. Besides, in this topic, we will discuss what is a millimetre of mercury and how to calculate mmHg? Millimetre of mercury is the short form for a millimetre of mercury.Īlso, it is a measuring unit that measures the pressure in the manometric unit. MmHg is certainly a very important calculation in Physics.
